The CF is one of the Portuguese institutions whose projects were chosen as recipients of this first edition, for their capacitation efforts at the level of clinical research.
According to Teresa Machado Luciano, vice-president of AICIB,
“These awards aim to support capacitation projects presented and developed by clinical research centres in hospitals of the Portuguese health system, in order to foster their organisation, professionalisation and boost their performance, thus increasing sponsor trust and the international recognition of Portuguese clinical research centres”.
Building capacity in clinical Research is a complex endeavour. Even more so if you not only want to build the capacity of an institution to collaborate in industry-sponsored trials organised internationally, but if you want to actually increase the capacity for clinicians and researchers to work together to turn important questions into running research projects that will eventually help their patients. This involves providing researchers access to knowledge and support in different areas, in order to foster creative solid projects and ensure that new studies and trials can progress swiftly through the administrative and regulatory evaluation that guarantee scientifically strong, ethically-sound research with the greatest impact in the lives of people.
Research on human health and behaviour is extremely resource-intensive. In particular, developing clinical studies often implies the integration of broadly different fields of medicine, basic and applied research, and the committed collaboration of multiple public and private stakeholders, meeting specific and stringent ethical and regulatory standards.
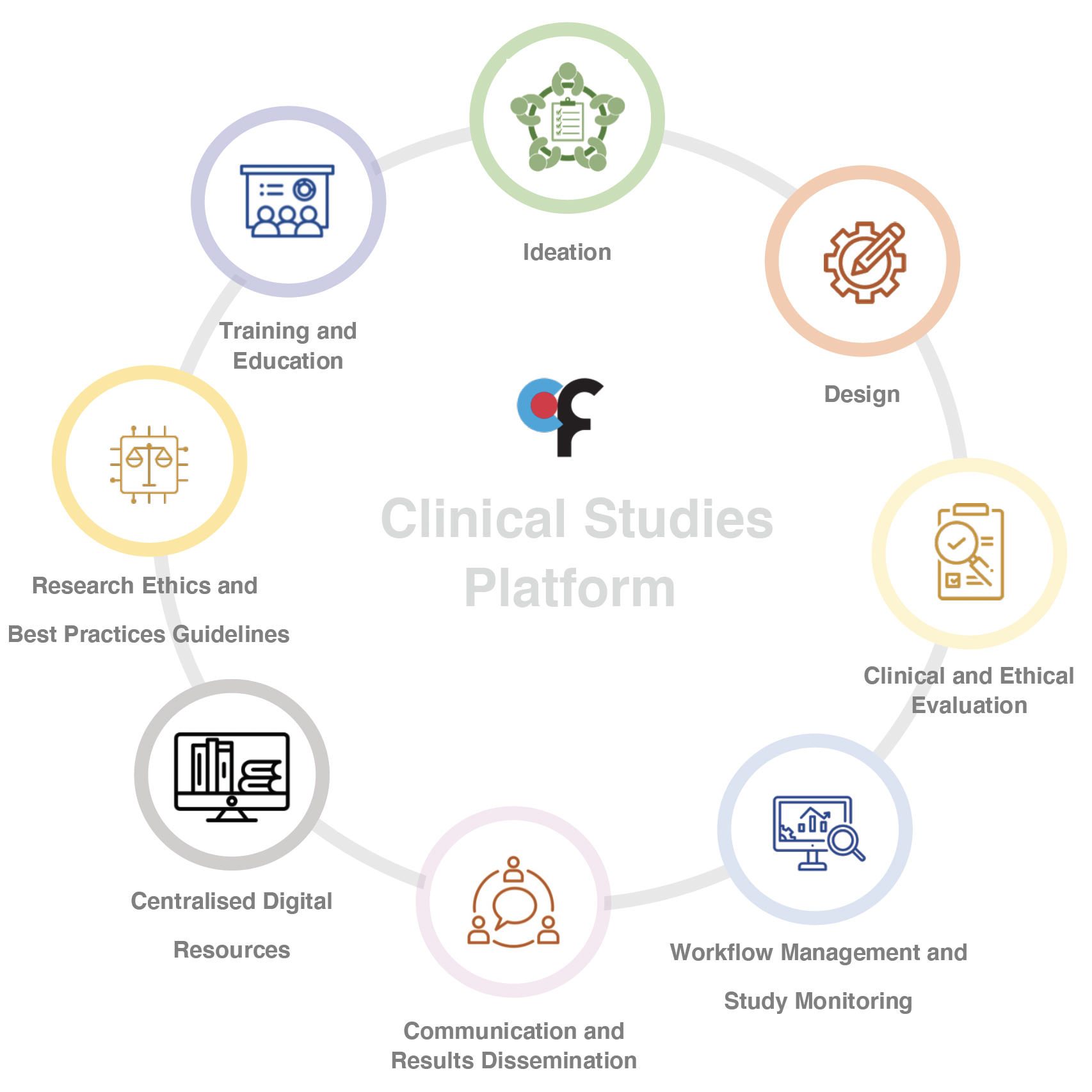
The CF Clinical Studies Platform aims to provide a structure to stimulate clinical research, integrating existing staffed support units with new digital resources, and introducing multi-stakeholder workgroups as a resource to improve research quality and the societal impact of both investigator and industry-driven projects.
This platform, formalised and accessible in digital form, goes beyond a digital tool: it brings together real people and organizes human resources and information, to promote and consolidate bridges between different stakeholders involved in the pursuit, ideation, preparation, and evaluation of clinical studies at the Champalimaud Centre for the Unknown (CCU).
Perhaps most importantly, this platform aims to promote better patient and public involvement in clinical research, with multi-stakeholder workgroups where researchers will be able to work with patients and patient advocates in the field of their clinical studies and interests. Partnerships with patient organisations and networks will bring patients and patient experts to meaningfully engage with researchers on the design of better research questions, protocols, patient information for informed consent, and communication of research results.
Clinical research that is more aligned with patients’ needs will consequently increase patient participation and recruitment, show greater and faster societal impact of research results and accelerate the adoption of new evidence into clinical practice.
By Ana Gerschenfeld, Health & Science Writer of the Champalimaud Foundation.


